By 2026, the standard medical practice of “prescribe and pray” has been technically retired. We have entered the era of Pharmacogenomics (PGx), a discipline that uses an individual’s genetic profile to dictate the exact drug and dosage required for optimal efficacy. In 2026, the variation in how two people respond to the same pill is no longer a medical mystery; it is a predictable data point. This shift has transformed the pharmacy from a dispensary into a center for Precision Pharmacokinetics.
The catalyst for this revolution was the “PGx-First” legislation enacted in early 2025 across several major health systems. This mandate technically requires a genomic screening before a physician can authorize prescriptions for high-risk or high-variability drug classes. In 2026, the goal is to eliminate the dangerous period of “trial and error”—where patients often spend months cycling through different antidepressants or blood thinners—by identifying the correct molecular match on day one.
This is not just about avoiding minor side effects; it is about systemic safety. In 2026, we recognize that what is a therapeutic dose for one person can be technically toxic for another due to their unique enzymatic architecture. By integrating genomic data directly into the electronic health record (EHR), the 2026 medical landscape has created a safety net that catches potential Adverse Drug Reactions (ADRs) before the prescription is even printed.
In 2026, prescribing a medication without a PGx panel is increasingly viewed as clinical negligence. We have moved from “educated guessing” to “molecular certainty,” saving billions in systemic costs and thousands of lives from adverse drug reactions.
Decoding the CYP450 Dashboard: Star Alleles and Metabolic Rates
The technical engine of PGx in 2026 is the Cytochrome P450 (CYP450) enzyme family in the liver. These enzymes are responsible for metabolizing the vast majority of clinical drugs. However, genetic variations—known as Star Alleles (e.g., CYP2D63*)—determine the speed and efficiency of this process. In 2026, your “CYP Dashboard” is a foundational part of your biological identity, telling doctors exactly how your liver will “process” the data of a new medication.
The efficiency of this metabolic process is quantified by the Pharmacological Clearance Rate (Cl), which measures the volume of plasma cleared of a drug per unit of time:
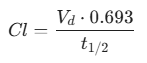
Where Vd is the volume of distribution and t1/2 is the drug’s half-life. In 2026, PGx allows us to adjust the dosage to maintain a steady-state concentration (Css) tailored to your specific Cl. To facilitate this, 2026 clinicians categorize patients into four distinct metabolic technical tiers:
- Poor Metabolizers (PM): Enzymes are nearly inactive; standard doses lead to toxic accumulation.
- Intermediate Metabolizers (IM): Reduced enzyme activity; requires downward dosage adjustment.
- Normal Metabolizers (NM): The “legacy” standard; drugs work as traditionally expected.
- Ultra-rapid Metabolizers (UM): Enzymes process the drug so fast it never reaches therapeutic levels; requires a higher dose or alternative medication.
High-Impact Areas: Mental Health, Cardiology, and Pain Management
In 2026, the impact of PGx is most visible in therapeutic areas where the “trial and error” method was historically most damaging. In Mental Health, for instance, the success rate of the first antidepressant prescribed has jumped from 30% to over 70% thanks to genomic matching of SSRIs and SNRIs. This technical precision has drastically reduced the “washout periods” and systemic despair associated with ineffective psychiatric treatment. The 2026 clinical guidelines now mandate PGx screening for five critical drug classes:
- SSRIs/Antipsychotics: To avoid serotonin syndrome or lack of response in PM/UM patients.
- Statins: To prevent SLCO1B1-related myopathy (muscle pain) in cardiovascular patients.
- Anticoagulants (Warfarin/Clopidogrel): To prevent life-threatening bleeding or stroke from incorrect dosing.
- Opioids: To identify patients who cannot convert codeine into morphine, rendering the pain relief technically zero.
- NSAIDs: To identify individuals at high risk for gastrointestinal bleeding or cardiovascular events.
The Digital Pharmacist: Real-Time Interaction Checks
The “Digital Pharmacist” of 2026 is an AI-integrated system that acts as a real-time genomic gatekeeper. When a doctor enters a prescription into an EHR, the system cross-references the drug’s metabolic pathway with the patient’s stored PGx Profile. If a mismatch is detected—such as a Poor Metabolizer being prescribed a standard dose of a high-risk drug—the system technically “locks” the order and suggests a genomic-safe alternative.
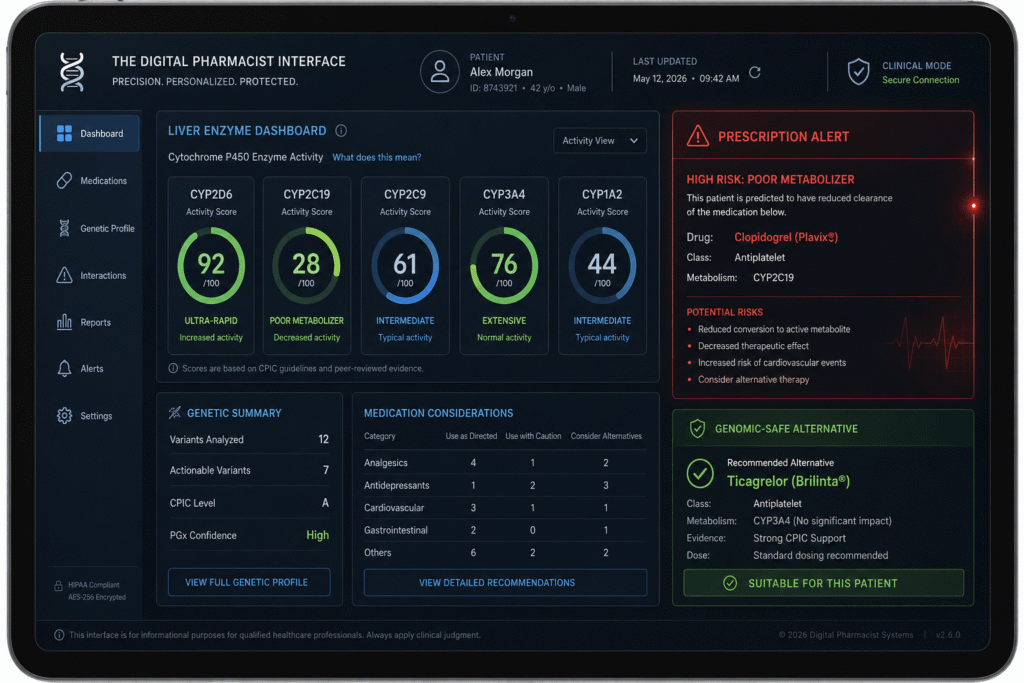
This infrastructure has effectively moved PGx from the laboratory to the bedside. In 2026, patients can access their “Prescription Compatibility Map” via their smartphones, allowing them to see which over-the-counter medications or supplements might technically interfere with their genetic predispositions. This level of transparency ensures that the patient is an active participant in their own Pharmacological Safety.
| Feature | Legacy Medicine (2020) | PGx Standard (2026) |
| Dosing Model | One-size-fits-all (Population avg) | Individual Genotype-based |
| Prescription Logic | Trial and Error | Predictive Molecular Matching |
| Side Effect Risk | High (Unpredictable) | Minimally Technical (Avoidable) |
| Efficacy Speed | Weeks/Months to verify | Immediate Optimal Dosing |
| Systemic Cost | High (Due to ADRs/Inefficacy) | Low (Due to Direct Success) |
FAQ: Navigating Your Genetic Prescription
Do I need to repeat my PGx test every year in 2026?
No. Unlike a blood sugar or cholesterol test, your germline DNA remains constant. A single, comprehensive Whole Genome PGx Panel performed in 2026 is technically valid for your entire life. However, as new drug-gene interactions are discovered, your Digital Health Record will automatically update your compatibility map without needing a new sample.
Is my genetic drug data private in 2026?
This is a primary technical concern. In 2026, PGx data is protected under strict Genetic Non-Discrimination laws. Reputable providers use On-Device Encryption, meaning your raw genetic data stays on your local device or a secured, air-gapped clinical server, and only the “Compatibility Result” (Pass/Fail/Adjust) is shared with the pharmacy.
Does insurance cover PGx testing in 2026?
Most major insurers in 2026 now cover 100% of the cost for an initial PGx panel. The actuarial data is clear: the one-time cost of the test is far lower than the cost of treating a single severe Adverse Drug Reaction or the long-term expenses of ineffective chronic disease management.
How does PGx affect my use of supplements and “Biohacking”?
PGx is essential for the 2026 biohacker. Many common supplements, like St. John’s Wort or high-dose Curcumin, technically induce or inhibit the same CYP450 enzymes as prescription drugs. Your PGx profile can tell you if a “natural” supplement will technically neutralize your medication or increase its toxicity.
Can PGx tell me if I will be allergic to a medication?
While PGx is excellent at predicting metabolic rates, it is only part of the “Allergy Equation.” In 2026, we use specific HLA (Human Leukocyte Antigen) markers within the PGx panel to identify risks for severe skin reactions (like Stevens-Johnson Syndrome). However, standard IgE-mediated allergies (like a penicillin rash) still require traditional clinical monitoring.
What is the future of “Gene Correction” for Poor Metabolizers?
By 2026, we have seen the first experimental trials using CRISPR-based Epigenetic Editing to “upregulate” enzyme production in Poor Metabolizers. While not yet a standard treatment, the technical goal is to one day “fix” a genetic metabolic deficit, allowing every patient to process medication as a Normal Metabolizer.